More than one million people worldwide live with cochlear implants — yet every single one of them must attach an external device to their head each morning before they can hear. That may be about to change. Envoy Medical’s Acclaim cochlear implant, now in a pivotal FDA clinical trial with all 56 participants activated, is targeting a Premarket Approval (PMA) submission to the FDA in Q2 2027. Here is what cochlear implant candidates, current CI users, and their families need to understand about this technology — and what remains uncertain.
What Is a Fully Implantable Cochlear Implant?
A traditional cochlear implant has two distinct parts: a surgically implanted receiver-stimulator beneath the skin, and an external sound processor clipped behind the ear or worn on a magnet. The external processor does the heavy lifting — it captures sound through a microphone, digitizes it, and transmits a signal through the skin to the internal implant. Without it, the user hears nothing.
A fully implantable cochlear implant places all components — microphone, processor, battery, and electrode array — entirely under the skin. There is no processor to put on in the morning, no battery pack to charge overnight, and no visible hardware marking you as a hearing device user. The concept is straightforward; the engineering challenge has taken decades.
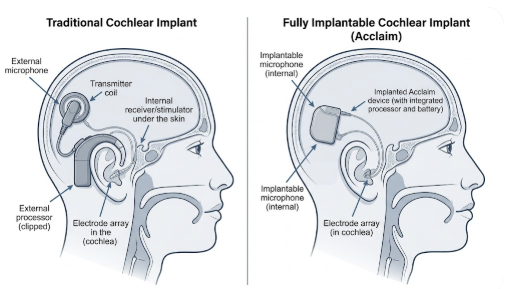
How the Acclaim CI Captures Sound Without a Microphone
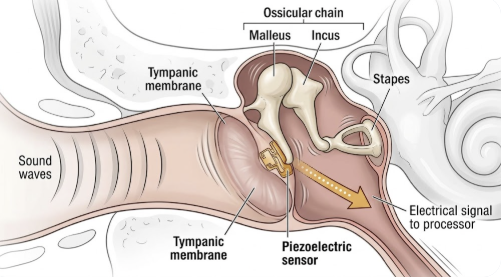
The central technical problem in building a fully implantable CI is sound capture. An external microphone benefits from sitting in open air at ear level, where sound waves arrive cleanly. Move that microphone under skin and bone, and performance degrades substantially — which is why earlier attempts at full implantation fell short.
Envoy Medical’s solution with the Acclaim is to skip the microphone entirely. Instead of capturing airborne sound waves, the Acclaim uses a proprietary piezoelectric sensor placed against the ossicular chain inside the middle ear. When sound enters the ear canal and causes the tympanic membrane and ossicles to vibrate, the sensor converts those mechanical vibrations directly into an electrical signal. The implanted processor then encodes this signal and delivers electrical stimulation to the cochlear electrode array — the same fundamental step that occurs in all cochlear implants.
The early feasibility data support this approach. In the first-in-human early feasibility study of three patients, all achieved hearing through the device at activation, and all became regular users over the follow-up period [Dornhoffer JR et al., Initial Experiences with the Envoy Acclaim® Fully Implanted Cochlear Implant, 2023]. One-year audiological follow-up confirmed preserved residual hearing in the implanted ear and electrical stimulation levels comparable to conventional CIs, though two of three patients added an ipsilateral hearing aid — in the same ear as the implant — to optimize speech perception scores [Dornhoffer JR et al., Early Hearing Outcomes and Audiological Experiences With a Novel Fully Implanted Cochlear Implant, 2024].
The Acclaim is not the only fully implantable CI in development. MED-EL’s Totally Implantable Cochlear Implant (TICI) used a different approach — integrating all audio processor components into the implant — and demonstrated comparable safety and speech performance to a conventional CI across 6 patients over 52 weeks [Lefèbvre PP et al., Rehabilitation of Human Hearing with a Totally Implantable Cochlear Implant, 2025]. The field is converging from multiple directions.
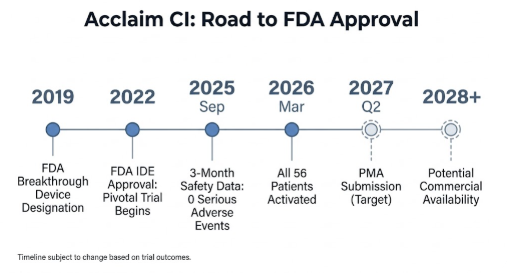
Where the Acclaim Trial Stands Right Now
The Acclaim received FDA Breakthrough Device Designation in 2019, which expedites development and review for devices that address unmet needs in serious conditions. In 2022, Envoy Medical received FDA approval to begin its pivotal clinical study — the final phase required before a PMA application.
As of March 2026, all 56 participants in the pivotal study have been implanted and activated. In April 2026, Envoy Medical presented the first podium data from the study: an interim analysis of the initial 10 patients at their six-month follow-up visits. The safety record to date has been clean — no serious adverse events or unanticipated device effects were reported through the three-month follow-up checkpoint, which prompted the FDA to approve expansion of the trial to its final stage.
Envoy Medical’s stated target is FDA PMA submission in Q2 2027. It is worth being precise about what that means: submission, not approval. FDA review of a PMA application typically takes 180 days at minimum, and additional review cycles are common. Commercial availability in the United States, if approved, would realistically begin no earlier than late 2028 or into 2029.
Fully Implantable vs. Traditional CI: A Direct Comparison
| Feature | Traditional Cochlear Implant | Acclaim CI (Investigational) |
|---|---|---|
| External processor required | Yes — worn daily | None |
| Water exposure | Limited; processor must be removed | Fully submerged use (by design) |
| Sleeping with device | Processor removed; no hearing during sleep | Continuous hearing (including sleep) |
| Sound capture mechanism | External microphone in air | Internal piezoelectric sensor (ossicular chain) |
| Battery management | External battery pack; daily charging | Transcutaneous charging through skin |
| MRI compatibility | Conditional (varies by device/field strength) | Under investigation |
| FDA status | Approved (multiple devices available) | Investigational — PMA submission Q2 2027 (target) |
| Surgical complexity | Standard CI procedure | More complex; requires middle ear sensor placement |
MRI compatibility deserves specific attention. With conventional CIs, MRI access is conditional — typically limited to 1.5T with magnet removal or specific protocol modifications, depending on the implant model. This is already a practical limitation for many CI users who need neuroimaging. Whether and how the Acclaim performs under MRI has not yet been publicly characterized from the pivotal trial data. It will be a defining question before broad adoption.
Clinical Perspective: What This Actually Changes for Patients
The following reflects my clinical perspective as an otolaryngologist, not a recommendation for any specific device or enrollment in any trial.
When patients ask about cochlear implants, the conversation almost always turns to lifestyle constraints within the first five minutes. Can I swim? Can I sleep normally? Will it show? The external processor is effective — but it is also a daily reminder of disability, and for many patients, that psychological burden is real.
A fully implantable CI does not improve the fundamental auditory outcome compared to a conventional device; the electrode array, stimulation parameters, and cochlear physiology are essentially identical. What changes is when the device works. Continuous access to sound — during sleep, in the shower, while swimming — is clinically meaningful. Patient-reported quality-of-life measures in the MED-EL TICI study showed improved satisfaction and comfort, consistent with what we would expect from eliminating the external hardware [Lefèbvre PP et al., Rehabilitation of Human Hearing with a Totally Implantable Cochlear Implant, 2025].
There are open clinical questions I watch carefully. Battery longevity under continuous use will determine how frequently patients need transcutaneous charging — and eventually, whether the internal battery requires surgical replacement. The early Acclaim feasibility data reported a 4-day average battery life before recharging [Dornhoffer JR et al., Early Hearing Outcomes and Audiological Experiences With a Novel Fully Implanted Cochlear Implant, 2024]. That is workable, but the long-term degradation curve over years matters enormously for a surgically implanted device.
A note on interpreting these early results: both Dornhoffer et al. studies were conducted with Envoy Medical funding, and the 2024 paper follows the same three-patient cohort first reported in 2023. These are not reasons to dismiss the findings, but they are reasons to hold them lightly until independent, larger-scale data from the pivotal trial becomes available.
Surgical complexity is also higher — placing a sensor on the ossicular chain requires middle ear access beyond standard CI implantation, which raises the skill threshold for surgeons and the recovery burden for patients. These are not arguments against the technology; they are the honest questions that any candidate should ask before enrollment or, eventually, elective surgery.
Who Is a Candidate?
The Acclaim pivotal trial enrolls adults with severe-to-profound bilateral sensorineural hearing loss (SNHL) who meet standard cochlear implant candidacy criteria — including a clear cochlear space and healthy cochlear nerve confirmed by MRI or CT. These criteria largely mirror what defines conventional CI candidacy today [Babajanian EE et al., Fully Implanted Cochlear Implants, 2024].
For patients who are already strong conventional CI candidates and are particularly motivated by the lifestyle advantages of full implantation, the Acclaim represents a compelling option — once approved. For patients with significant comorbidities, prior middle ear surgery, or complex cochlear anatomy, the more involved surgical approach will require individualized assessment.
The Acclaim is currently available only to trial participants at designated investigational sites in the United States. There is no pathway for general access until FDA approval, and international regulatory submissions would require separate applications following any US approval.
Key Takeaways
- The Acclaim cochlear implant by Envoy Medical is a fully implanted CI designed without any externally worn components — no external processor, microphone, or battery pack.
- It captures sound using a piezoelectric sensor placed on the ossicular chain inside the middle ear, eliminating the need for an external microphone.
- As of March 2026, all 56 patients in the pivotal clinical trial have been activated; the first interim data (initial 10 patients at six months) was presented in April 2026 with no serious adverse events reported.
- Envoy Medical targets FDA PMA submission in Q2 2027; commercial availability in the US, if approved, would not be expected before 2028–2029.
- MRI compatibility, long-term battery life, and surgical complexity remain open questions that will shape clinical adoption.
- The device holds FDA Breakthrough Device Designation, awarded in 2019.
FAQ
Is the Acclaim cochlear implant FDA approved?
No. As of 2026, the Acclaim CI is an investigational device. Envoy Medical completed enrollment of its pivotal clinical trial in early 2026 and targets FDA PMA submission in Q2 2027 — but submission is not approval, and regulatory review typically adds 6–18 additional months before any commercial access.
How is a fully implantable cochlear implant different from a traditional one?
Both deliver electrical stimulation to the cochlea via an implanted electrode array. The difference is the sound pathway: traditional CIs rely on an external processor worn on the head to capture and transmit sound, while a fully implantable CI like the Acclaim captures sound through an internal sensor and processes it entirely beneath the skin. The surgical outcome and cochlear mechanics are comparable; the lifestyle experience is fundamentally different.
Can I swim or sleep with the Acclaim CI?
That is the central design advantage. Because no external components are worn, the device would allow continuous hearing during sleep, bathing, and water activities — a significant departure from conventional CIs. These specifications remain subject to final FDA-cleared labeling and have not yet been formally confirmed through the pivotal trial’s primary endpoints.
Who manufactures the Acclaim cochlear implant?
Envoy Medical, Inc., based in White Bear Lake, Minnesota, USA.
When could the Acclaim be available outside the United States?
International availability depends on separate regulatory submissions to agencies such as the CE Mark authority in Europe or the MFDS in South Korea. No timeline for international submissions has been announced as of mid-2026.
Joonpyo Hong, MD is a board-certified otolaryngologist practicing in Korea. This article reflects his clinical interpretation of published research and does not constitute individual medical advice.
References
- Dornhoffer JR, Lawlor SK, Saoji AA, Driscoll CLW. Initial Experiences with the Envoy Acclaim® Fully Implanted Cochlear Implant. J Clin Med. 2023;12(18):5875.
- Dornhoffer JR, DeJong MD, Driscoll CLW, Saoji AA. Early Hearing Outcomes and Audiological Experiences With a Novel Fully Implanted Cochlear Implant. Otol Neurotol. 2024;45(10):e727–e734.
- Lefèbvre PP, Müller J, Mark G, et al. Rehabilitation of human hearing with a totally implantable cochlear implant: a feasibility study. Commun Med (Lond). 2025;5(1):10.
- Babajanian EE, Dornhoffer JR, Driscoll CLW. Fully Implanted Cochlear Implants. Curr Otorhinolaryngol Rep. 2024;12:61–65.
- Envoy Medical. Envoy Medical Announces Milestone as All 56 Patients Activated in Acclaim® Cochlear Implant Trial. Hearing Health & Technology Matters. 2026. Available at: https://hearinghealthmatters.org/hearing-news-watch/2026/envoy-acclaim-trial-activation/
- Envoy Medical. Envoy Medical Highlights First Clinical Data Presentations from Pivotal Clinical Study of Fully Implanted Acclaim® Cochlear Implant. Press release. April 2026. Available at: https://ir.envoymedical.com/news-events/press-releases/detail/90/envoy-medical-highlights-first-clinical-data-presentations-from-pivotal-clinical-study-of-fully-implanted-acclaimr-cochlear-implant
- Cochlear Ltd. Cochlear Announces Availability of Nucleus Nexa System to US Military Veterans. Press release. January 8, 2026. Available at: https://www.cochlear.com/us/en/corporate/media-center/media-releases/2026/cochlear-announces-availability-of-nucleus-nexa-system-to-us-military-veterans