A smartphone AI otoscope promises something specific: attach a $40 scope to a phone, record three seconds of video of a crying toddler’s eardrum, and the app returns a verdict — acute otitis media, 94% confidence. A pediatrician, on a good day, is right somewhere between 50% and 70% of the time. That gap is no longer hypothetical — two peer-reviewed studies, one in 2024 and one in 2026, have now measured it.
This article walks through what those smartphone AI otoscope studies actually found, why physicians miss so many ear infections in the first place, where the AI is genuinely useful and where it falls short, and why Korean families cannot legally use any of these tools today.
How Accurate Is AI for Diagnosing Otitis Media?
In March 2024, a team from UPMC and the University of Pittsburgh published the first large clinical validation of a smartphone AI otoscope in JAMA Pediatrics (Shaikh et al., 2024). The researchers trained two deep-learning models on 1,151 tympanic membrane videos from 635 children seen at outpatient UPMC pediatric offices between 2018 and 2023. On the held-out test set, the tool hit ~94% sensitivity and ~94% specificity — in layman’s terms, about 94% accuracy — for distinguishing acute otitis media (AOM) from other conditions.
Two years later, a Chinese engineering group pushed the same smartphone AI otoscope concept onto far cheaper hardware. Their embedded system, published in Acta Otolaryngologica (Lv et al., 2026), trained on 19,522 tympanic membrane images and ran inference directly on an STM32H7-class microcontroller — the kind of chip used in consumer electronics that costs under five dollars. It classified normal tympanic membranes, AOM, and otitis media with effusion (OME) with above 90% sensitivity and specificity — in layman’s terms, above 90% accuracy.
For comparison, the clinician baseline is sobering. Multiple studies of primary care doctors and pediatric residents report diagnostic accuracy for AOM ranging from 30% to 84% (Pichichero, reviewed in CIDRAP coverage of the 2024 UPMC release). The floor of that range is worse than a coin flip on a condition that generates millions of antibiotic prescriptions a year.
Why Do Doctors Miss Ear Infections So Often?
The tympanic membrane (eardrum) is a small, angled target roughly the size of a pencil eraser (about 1×1 cm, 0.1 mm thick). In a cooperative adult, doctors can get enough time for inspection. In a frightened two-year-old, you get three seconds — and half the view is obscured by cerumen, a canal that twists, or a child who turns their head the moment the speculum touches skin (which triggers sharp pain, crying, and the end of cooperation).
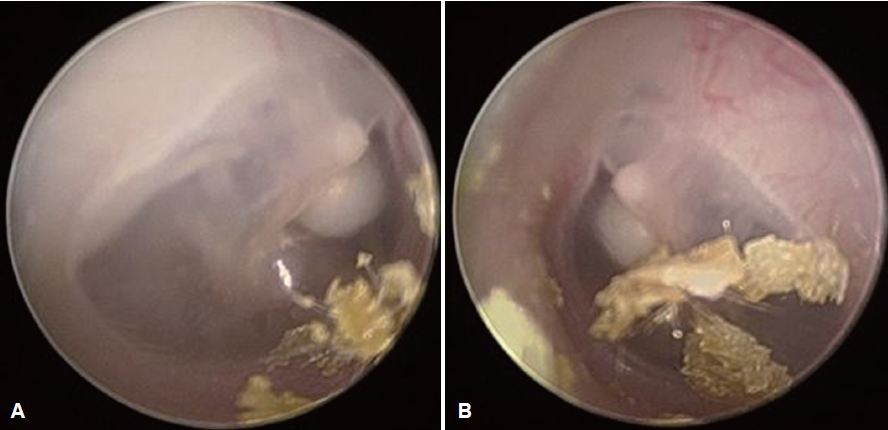
[This is a photo of the tympanic membrane from a 3-year-old boy with bilateral congenital cholesteatoma, excerpted from a case report I previously authored. I remember the absolute struggle to get this shot—holding the child’s head still and practically pleading with him to give me just three seconds.]
Diagnostic accuracy for AOM depends heavily on experience. Although residents and specialists use the same billing codes, their exam quality varies significantly. This discrepancy leads to misdiagnosis, which fuels antibiotic overuse for viral infections. Unnecessary prescriptions offer no benefit, increase resistance, and cause adverse reactions. Improved diagnostic tools are therefore essential for antibiotic stewardship.
Clinical Perspective — Personal Take
Sometimes a bulging, opaque membrane has been called “normal red from crying.” Sometimes a retracted but intact membrane with a small effusion has been treated with a ten-day amoxicillin course that was never indicated.
What makes AI plausible here is that the specific failure mode of human otoscopy — brief, uneven view of a small target under time pressure — is exactly the problem computer vision handles well. A thirty-frame video gives the model thirty looks instead of one. Lighting is consistent. The algorithm does not get tired at 5 p.m. on a Friday, and it does not adjust its threshold because a distressed parent is asking for antibiotics.
What AI cannot do is the rest of the visit. It cannot palpate a mastoid, ask whether the fever started before or after the ear pain, weigh a six-month recurrence history against a first episode, or decide whether a child needs tympanostomy tubes. The correct frame for every tool in this class is augmentation — a second reader that flags uncertainty and says “please see a clinician” when it is not sure. The UPMC paper itself frames the tool this way.
Can I Use a Smartphone AI Otoscope at Home Right Now?
United States
Not yet, at least not with clinical validation. The UPMC tool remains in a research and regulatory pipeline; no FDA-cleared consumer smartphone AI otoscope is currently on the U.S. market as of early 2026. Several consumer smartphone otoscopes are sold without AI — they produce an image, and the interpretation is left to whoever looks at it. Image quality varies widely, and there is no reason to assume a parent untrained in otoscopy will interpret that image better than a pediatrician.
South Korea
The situation is more constrained. Telemedicine reimbursement in South Korea remains narrow, and remote pediatric diagnosis based on a parent-captured image is not a covered or clearly regulated pathway. Korea’s MFDS has an AI medical device approval track, but pediatric diagnostic AI for home use is not yet a standard category with clearance. Korean parents who want a smartphone AI otoscope today have no legal, reimbursed route to access it — a policy gap worth watching as domestic medical AI vendors mature.
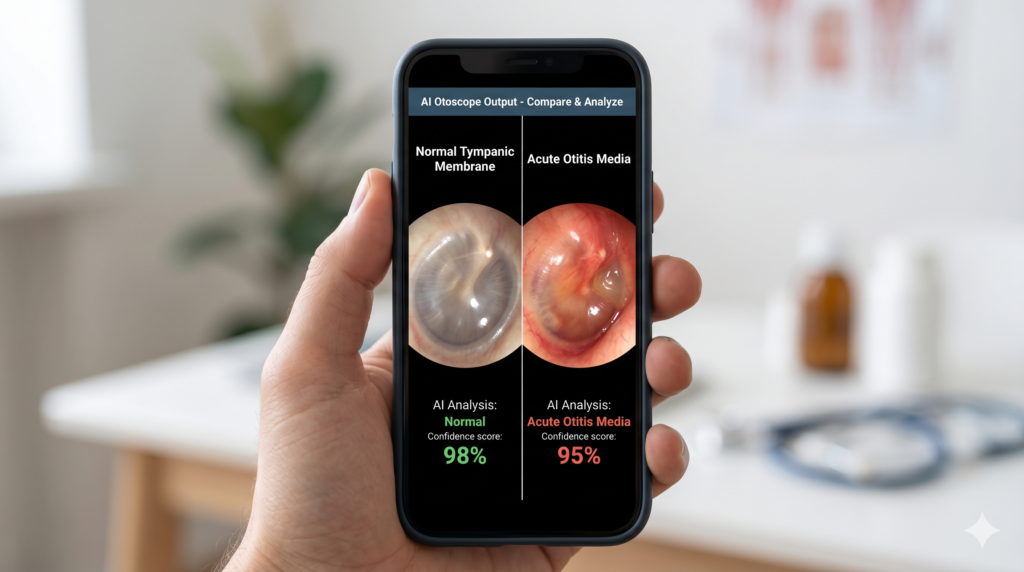
[Smartphone AI otoscope output comparing acute otitis media versus normal tympanic membrane with confidence scores (AI photo)]
Key Takeaways
- Smartphone AI otoscopes achieved over 90% accuracy for pediatric acute otitis media in recent peer-reviewed studies.
- Clinician accuracy for the same diagnosis ranges from 30% to 84%, depending on training and setting.
- The UPMC model analyzed short eardrum videos from 635 children; the 2026 embedded model runs on microcontroller hardware costing under five dollars.
- Improved diagnostic accuracy is positioned as an antibiotic stewardship tool, with the potential to reduce unnecessary pediatric prescriptions.
- No smartphone AI otoscope is FDA-approved for consumer home use as of early 2026, and Korean telemedicine regulations currently block patient-facing deployment.
- Clinical judgment — pain assessment, fever timing, recurrence history, surgical candidacy — remains the domain of the physician. AI belongs in the visit as a second reader, not a replacement.
FAQ
Can a smartphone really diagnose my child’s ear infection?
According to a handful of recent studies, possibly — with over 90% accuracy across the two largest published reports. No smartphone AI otoscope is yet FDA-approved for consumer home diagnosis. The technology may be ready for use; regulatory clearance and reimbursement pathways are certainly not.
Is AI more accurate than my pediatrician?
In controlled studies, the AI matched or exceeded the published clinician range of 30–84% accuracy. This does not mean AI should replace your doctor. Clinical context — fever timing, ear pain quality, recurrence pattern, and examination of the rest of the child — still drives the treatment decision. Clinical context, however, is never a workaround for a direct otoscopic exam.
When will a smartphone AI otoscope be available to parents?
FDA clearance in the United States and MFDS approval in Korea are the rate-limiting steps. Based on typical device review timelines, a cleared consumer smartphone AI otoscope is likely one to three years out, with institutional and primary care deployment probably arriving first.
Will this reduce antibiotic overuse in children?
That is the explicit design goal, and the math supports it. If a meaningful fraction of current AOM misdiagnoses lead to unnecessary antibiotic prescriptions, even a modest accuracy improvement at the point of diagnosis translates into fewer prescriptions, less resistance pressure, and fewer adverse drug reactions in children.
Medical disclaimer: This article is for educational purposes and does not constitute medical advice. If your child has ear pain, fever, or persistent symptoms, see a qualified clinician.
Joonpyo Hong, MD is a board-certified otolaryngologist practicing in Korea. This article reflects his clinical interpretation of published research and does not constitute individual medical advice.
Sources
- Shaikh N, Conway SJ, Kovacevic J, et al. Development and Validation of an Automated Classifier to Diagnose Acute Otitis Media in Children. JAMA Pediatr. 2024;178(4):401–407.
- Lv C, Jia D, Gao B, Xia L, Wang X, Wu Z. Deep learning-assisted otoscopic screening for paediatric otitis media: feasibility of edge deployment. Acta Otolaryngol. 2026 Mar 30:1–9.
- Pichichero ME. Diagnostic accuracy, tympanocentesis training performance, and antibiotic selection by pediatric residents in management of otitis media. Pediatrics. 2002;110(6):1064–70.
- CIDRAP. AI tool shows high accuracy for diagnosing ear infections. 2024.