It is estimated that approximately 15 million people worldwide have experienced persistent olfactory dysfunction lasting more than 6 months following COVID-19 [Chua TB, 2022] — and clinicians still rely on patients sniffing paper strips to measure it. Surely, someone must have thought about AI electronic noses. A research team in South Korea recently published an idea: an artificial intelligence-powered electronic nose that distinguishes nine scent molecules with accuracy approaching the human olfactory system. What does this actually mean for ENT practice?
How Does an Electronic Nose Work?
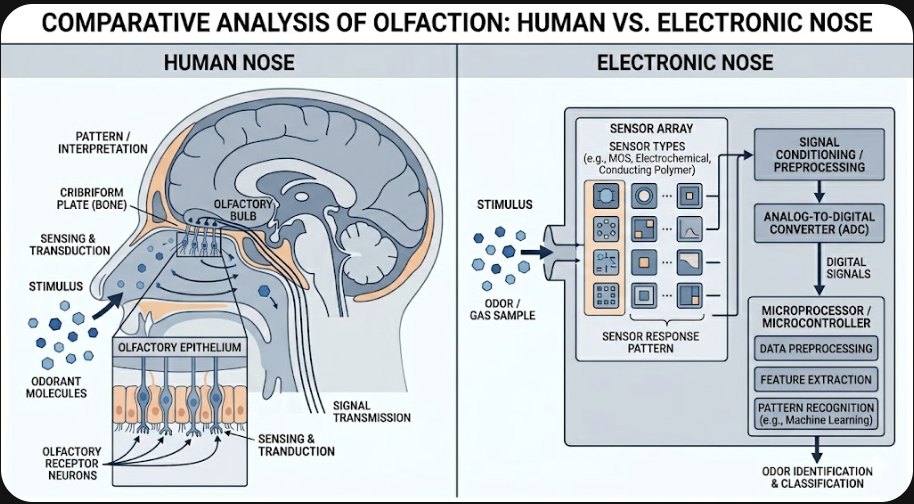
The human nose detects smell through roughly 400 types of olfactory receptors. No single receptor is dedicated to a single scent. Instead, each odor activates a unique combination of receptors — a process called combinatorial coding — and the brain reads the pattern as a specific smell.
An electronic nose (e-nose) mimics this logic. A sensor array responds to volatile organic compounds (VOCs) in the air, converting chemical signals into electrical ones. A machine learning algorithm then analyzes the pattern of responses across multiple sensors to classify the odor. The more diverse the sensors, the richer the pattern — and the more odors the system can distinguish.
The 2025 DGIST study took this a step further. Researchers fabricated a porous laser-induced graphene (LIG) array doped in situ with cerium oxide (CeO₂) nanoparticles. By varying the laser irradiation parameters during fabrication, they produced ten sensors with meaningfully different chemical and physical properties — in a single manufacturing step. The array successfully classified nine representative odorants including geraniol (floral), d-limonene (citrus), eugenol (spicy), and 2,3,5-trimethylpyrazine (nutty), among others [Lim H, 2025].
The manufacturing efficiency here matters. Most e-nose sensor arrays require complex, repetitive fabrication of individually distinct sensors. The DGIST approach achieves sensor diversity through one-step laser patterning — a step toward the kind of cost reduction that medical devices require.
E-Nose vs. Standard Smell Tests: An Honest Comparison
Before getting excited about e-noses in ENT clinics, it helps to understand what current smell testing actually does.
| Sniffin’ Sticks / UPSIT | AI E-Nose | |
|---|---|---|
| What it measures | Patient’s subjective olfactory experience | VOC concentration and pattern in air |
| Output | Threshold, Discrimination, Identification scores | Odor classification |
| Strengths | Validated, inexpensive, widely available | Objective, reproducible, not examiner-dependent |
| Current clinical status | Standard of care | Research stage |
The Sniffin’ Sticks TDI test and the University of Pennsylvania Smell Identification Test (UPSIT) remain the workhorses of olfactory evaluation. They are validated, inexpensive, and capture what patients actually experience. An e-nose, by contrast, measures the chemistry of the air — not the brain’s interpretation of it.
This is not a trivial distinction. A patient with parosmia smells things differently, not just more faintly. No e-nose currently on the table addresses qualitative distortions like that. The tools measure different things, and for now, that comparison favors the established tests for clinical diagnosis.
What Medical Problems Could an AI E-Nose Actually Help With?
There are several application scenarios where e-nose technology has genuine, near-term clinical potential — none of which are “replacing the ENT consultation.”
Objective monitoring of olfactory training outcomes. Olfactory training — repeated daily exposure to four or more scents — is currently the best-supported non-pharmacological intervention for post-viral smell loss. Monitoring its effectiveness relies almost entirely on patient self-report and psychophysical testing. A small, calibrated e-nose that measures VOC detection thresholds consistently over weeks could provide a more objective response curve.
Safety assistance for severe anosmia. Patients who completely lose their sense of smell cannot detect natural gas, smoke, or spoiled food. Harvard otolaryngologist Eric Holbrook has noted that a miniaturized e-nose worn in or near the nose could serve as a hazard-alert device for this population — not to restore smell, but to compensate for the most dangerous gap it leaves (Stanley S, Science, 2025).
Breath-based VOC biomarker detection. The broader literature on e-nose in medicine focuses on exhaled air analysis for systemic diseases — lung cancer, Helicobacter pylori infection, and diabetes have all been explored. For ENT specifically, applications in sinonasal infection and chronic rhinosinusitis are early but plausible.
Routine olfactory diagnosis in ENT clinics is not realistic in the near term. The DGIST sensor has been tested on nine pure odorant molecules under laboratory conditions. Real-world air contains hundreds of competing VOCs. Getting from nine laboratory odors to a clinical-grade olfactory screening device is a substantial engineering and regulatory journey.
Where Does AI in Rhinology Actually Stand?
A 2023 systematic review covering AI applications in rhinology identified 39 eligible studies — most of them technical papers demonstrating AI model feasibility rather than clinical validation [Bulfamante AM, 2023]. The same pattern applies to olfactory AI: the research is accelerating, but clinical translation remains thin.
This is not a criticism of the science. It reflects the standard gap between proof-of-concept and regulatory-grade medical device. For context, Sniffin’ Sticks took years of psychometric validation work before becoming widely accepted. An objective AI-based olfactory tool will require the same kind of investment in normative data, test-retest reliability, and clinical outcome correlation.
The encouraging signal is that the hardware problem — making a sensitive, diverse, low-cost sensor array — is getting solved. The DGIST 2025 paper advances that frontier meaningfully.

Clinical Perspective
The following reflects a clinical interpretation of the published research and does not represent a systematic review of the literature. The opinions expressed are those of the author.
Olfactory complaints are among the more diagnostically frustrating presentations in ENT — not because the pathology is obscure, but because the measurement tools are inherently subjective. Post-COVID hyposmia, sinusitis-related smell loss, and qualitative distortions like parosmia and phantosmia all rely heavily on patient self-report and psychophysical testing. The core problem is the subjectivity gap: when a patient reports that their smell has “improved a little” after three months of olfactory training, it is genuinely difficult to know whether that reflects true peripheral recovery, central adaptation, or the placebo component of an active intervention.
This is where an objective, reproducible measure of olfactory sensitivity — even an imperfect one — would add clinical value. The most credible near-term application for AI e-nose technology in ENT is not first-visit diagnosis, but longitudinal monitoring of patients already under management. For that purpose, absolute calibration precision matters less than measurement consistency across repeated sessions. A device that reliably tracks directional change over weeks would be useful, even if it cannot map precisely to psychophysical thresholds.
The wearable hazard-detection scenario deserves equal attention — not as a diagnostic device, but as assistive technology. Severe anosmia carries a daily safety burden that is underappreciated in clinical settings. Patients cannot detect gas leaks, smoke, or spoiled food. A miniaturized e-nose that alerts to a small set of high-priority hazard odors would address the most concrete practical risk of smell loss, even without capturing any of the richness of normal olfaction.
What the 2025 ACS Nano paper does not represent is evidence that AI-based diagnosis of smell disorders is clinically imminent. It is a well-executed materials science result demonstrating sensor feasibility under laboratory conditions. The translational gap between a controlled nine-odor classification task and a clinical-grade olfactory screening tool is substantial — and in olfactology, technically promising tools have stalled at that gap before.
Key Takeaways
- An electronic nose (e-nose) converts airborne volatile organic compounds into electrical signals and uses AI pattern recognition to classify odors, modeling how the human olfactory system uses combinatorial receptor coding.
- In 2025, Lim H et al. (ACS Nano) demonstrated a laser-induced graphene sensor array with cerium oxide nanoparticles that successfully classified nine distinct odorant molecules using machine learning.
- Current standard olfactory tests (Sniffin’ Sticks, UPSIT) measure the patient’s subjective sensory experience; e-nose technology measures the chemistry of air — the two approaches are complementary, not interchangeable.
- The most credible near-term clinical use for AI e-nose in ENT is objective longitudinal monitoring of olfactory training outcomes, not first-visit diagnosis.
- E-nose technology for medical use remains in the research stage; clinical adoption requires large-scale validation, standardization, and regulatory approval.
FAQ
How does an electronic nose work for medical diagnosis? An e-nose uses an array of gas sensors to detect volatile organic compounds, converts the signals into electrical patterns, and applies machine learning to classify them. In medical contexts, the target is usually a characteristic VOC profile associated with a disease state or — in olfactory research — specific odorant molecules that represent different scent categories.
Can AI replace smell tests in ENT clinics? No, not at this stage. Standard psychophysical tests like Sniffin’ Sticks measure what patients actually smell, which requires the patient. An e-nose measures the chemistry of air, which provides objective data but does not capture qualitative experiences like parosmia or phantosmia. The tools address different questions, and current e-nose technology is not validated for clinical olfactory diagnosis.
What is an e-nose used for in healthcare today? The most developed medical e-nose applications are in breath analysis for systemic diseases — lung cancer screening, H. pylori detection, and metabolic disorders have been studied. Olfactory-specific clinical applications in ENT are earlier stage, though monitoring of olfactory training and hazard detection for anosmic patients are both technically plausible near-term uses.
Will an electronic nose help people with anosmia? Potentially, in two ways: as a wearable hazard-detection device (alerting to gas leaks, smoke, or spoiled food that the patient cannot smell), and as a monitoring tool to track whether therapeutic interventions are improving olfactory function over time. It will not restore the lived experience of smell. ENT specialists like Eric Holbrook at Harvard have described a miniaturized wearable e-nose as one realistic near-term use case for this population.
References
- Lim H, Kwon H, Jang JE, Kwon HJ. Intelligent Olfactory System Utilizing In Situ Ceria Nanoparticle-Integrated Laser-Induced Graphene. ACS Nano. 2025 May 13;19(18):17850-17862.
- Bulfamante AM, Ferella F, Miller AM, Rosso C, Pipolo C, Fuccillo E, Felisati G, Saibene AM. Artificial intelligence, machine learning, and deep learning in rhinology: a systematic review. Eur Arch Otorhinolaryngol. 2023 Feb;280(2):529-542.
- Chua TB, Son D, Wee J, et al. Trajectory of smell and taste impairment in patients with COVID-19: a 6-month longitudinal study. BMJ. 2022;378:e071407.
- Stanley S. Artificial ‘nose’ tells people when certain smells are present. Science. 2025 Nov 25. Available from: https://www.science.org/content/article/artificial-nose-tells-people-when-certain-smells-are-present
Joonpyo Hong, MD is a board-certified otolaryngologist practicing in Korea. This article reflects his clinical interpretation of published research and does not constitute individual medical advice.